There is only one envelope protein Gp on thogotovirus surface which shows no obvious homology with the envelope proteins of influenza viruses. Previous studies showed that THOV Gp can induce hemagglutination, but lacks neuraminidase or esterase activity. The evidence implies that the entry and release mechanisms of thogotoviruses are substantially different from that of influenza viruses.
In addition, the receptors on host cell and entry pathways of thogotoviruses are yet unclear. Therefore, studies on the entry, transmission, and evolution of these viruses are of great significance for prevention and control of related diseases and potential epidemics.
Professor GAO Fu (George Fu Gao)’s group from Institute of Microbiology of Chinese Academy of Sciences determined the crystal structures of THOV and DHOV Gps at atomic resolutions. Both structures display typical characteristics of class III viral fusion proteins in postfusion conformation (Figure 1).
Compared with other class III members, including vesicular stomatitis virus Gp, herpes simplex virus gB, and baculovirus Gp64 proteins, these structures were found to share highly similar domain architectures, especially to baculovirus Gp64. Among the five domains of Gp, domain I is the most conserved region, in particular the fusion loops at the bottom. Domain II, however, shows the highest variability which may contribute to the different host tropisms of these viruses.
Besides, the structures of THOV and DHOV Gps harbor a pH-sensitive interface in the center of domain III, where many charged residues locate to form extensive charge-charge interaction networks that may induce significant conformational changes in response to environmental pH shifts. This hypothesis was further confirmed by circular dichroism studies on Gps at different pHs. These observations together imply that thogotoviruses may enter host cells through the endocytosis pathway and the membrane fusion process might occur in the endosome.
Despite the similarity in overall structures, THOV/DHOV Gp and baculovirus Gp64 show obvious local structural differences due to domain shifts between each other. In addition, these proteins display distinct surface electrostatic potential patterns with a certain propensity of transition. The surface of baculovirus Gp64 is dominated by negative charges with a few discrete positive-charged areas, whereas DHOV Gp is rich in positive charges and THOV Gp is in between with some local features more similar to baculovirus Gp64 (Figure 2).
These similarities and differences in structure demonstrate that thogotovirus Gps and baculovirus Gp64s are of the same origin in evolution but diverged in different routes. Baculovirus Gp64 evolves in adaptation to insect hosts, while THOV and DHOV Gps evolve to favor mammalian hosts. In addition, the evolving routes of THOV Gp and DHOV Gp also show obvious difference, which enables THOV Gp to retain some features of baculovirus Gp64. To further testify the theory, researchers conducted phylogenic analysis on thogotovirus Gp homologs which also showed a similar trend of evolution and perfectly supported the hypothesis.
These findings would intensify the understanding on the pathway of thogotovirus entry and enrich our knowledge on the origin and evolution of all members of Orthomyxoviridae, which is of great theoretical and practical importance for prevention and control of related viral diseases and epidemics. The study was published in PNAS.
The study was granted by the Strategic Priority Research Program of the Chinese Academy of Sciences, the China Ministry of Science and Technology National 973 Project, National Natural Science Foundation of China, and the Excellent Young Scientist Program and the Youth Innovation Promotion Association of the Chinese Academy of Sciences.
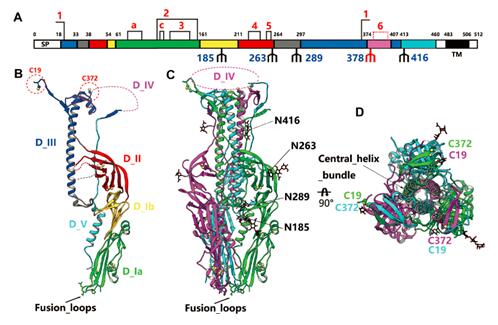
Figure 1. Overall structure of THOV Gp (Image by Prof. Gao’s group)
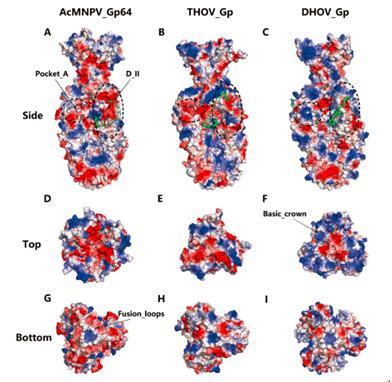
Figure 2. The surface electrostatic potential patterns of THOV/DHOV Gp and baculovirus Gp64 (Image by Prof. Gao’s group)
