The Zika virus (ZIKV) nonstructural protein 1 (NS1) functions in viral replication, pathogenesis, and immune evasion. George F. Gao and Yi Shi from Institute of Microbiology Chinese Academy of Sciences in Beijing and their colleagues solved the crystal structure of full‐length NS1 protein from the ZIKV involved in the 2015 outbreak in Brazil, and found an extended membrane association interface contributed by the hydrophobic “spike” of a long intertwined loop, providing important information for ZIKV pathogenesis and development of diagnostic tools.
The article entitled “Contribution of intertwined loop to membrane association revealed by Zika virus full‐length NS1 structure” was published in The EMBO Journal recently
(http://emboj.embopress.org/content/early/2016/08/30/embj.201695290).
The association of ZIKV infections with microcephaly and neurological diseases has highlighted an emerging public health concern. ZIKV NS1, a major host‐interaction molecule that functions in flaviviral replication, pathogenesis, and immune evasion. Of note, a long intertwined loop is observed in the wing domain of ZIKV NS1, and forms a hydrophobic “spike”, which can contribute to cellular membrane association. For different flaviviruses, the amino acid sequences of the “spike” are variable but their common characteristic is either hydrophobic or positively charged, which is a beneficial feature for membrane binding. Comparative studies with West Nile and Dengue virus NS1 structures reveal conserved features, but diversified electrostatic characteristics on both inner and outer faces.
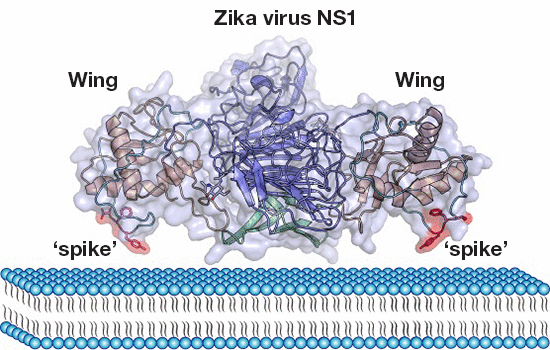
Contribution of intertwined loop to membrane association revealed by Zika virus full-length NS1 structure (Image by Prof. Gao’s group).
The team suggests that different mechanisms of flavivirus pathogenesis and should be considered during the development of diagnostic tools.
