Scientists reveal cholesterol as key driver of
impaired innate-adaptive immune axis in tuberculosis granulomas
As a major global medical challenge, chronic lung infections are associated with compromised local immunity that enables long-term pathogen survival. In response to persistent pathogenic stimuli, host immune cells typically aggregate to form granulomas. These structures share features with the tumor microenvironment, playing a central role in determining infection outcomes. However, the specific immunological features and regulatory mechanisms in infectious granulomas are not fully understood.
Recently, Prof. LIU Cui Hua’s group at the Institute of Microbiology of the Chinese Academy of Sciences (IMCAS), in collaboration with Profs. WANG Jing and WANG Shuo from IMCAS and PANG Yu from Beijing Chest Hospital, has revealed the spatiotemporal transcriptional landscapes of key immune cell populations in tuberculous granulomas and elucidated a previously unrecognized mechanism by which cholesterol accumulation disrupts the innate-adaptive immune interaction in granulomas (Figure 1). This work was published in Nature Microbiology.
Tuberculosis (TB), a major lung infection caused by Mycobacterium tuberculosis (Mtb), is one of the most frequent cause of granulomas. Currently, TB has re-emerged as the world’s leading infectious killer. This urgent situation prompts the development of novel host-directed therapies. However, progress is hampered by a limited understanding of TB immunology.
By employing multiplex immunohistochemistry and spatial transcriptomics on human specimens and mouse models, the research team systematically mapped the spatial distribution, transcriptional profiles, and intercellular interactions of macrophages and T cells within TB granulomas. They revealed that the immune heterogeneity of these two cell populations is shaped by multiple factors, including the tissue pathology (necrotic or non-necrotic granulomas), immune cell location (inner or peripheral regions of the granulomas), proximity to specialized structures (multinucleated giant cells or tertiary lymphoid structures), intracellular bacterial persistence (Mtb-infected or non-infected cells), and the infection stage.
Through comparative analysis with the lung tumor microenvironment, the team further identified a unique phenotypic signature in TB granulomas, an impaired macrophage, CD4+ T cell interaction closely associated with lipid accumulation. Mechanistically, they found that Mtb can induce lysosomal cholesterol accumulation in macrophages via its components such as mycolic acids. This leads to the retention of MHC-II within lysosomes and subsequent impairment of antigen presentation. This impairment compromises Mtb-specific CD4+ T cell responses and results in dysfunctional anti-Mtb immunity in granulomas. Notably, pharmacologically restoring lung cholesterol homeostasis can enhance host protective immunity against Mtb in mice.
Collectively, these findings broaden the understanding of the heterogeneity and plasticity of regional immunity within the lung infectious microenvironment, and propose a cholesterol-targeting strategy for restoring innate-adaptive immune interactions against TB.
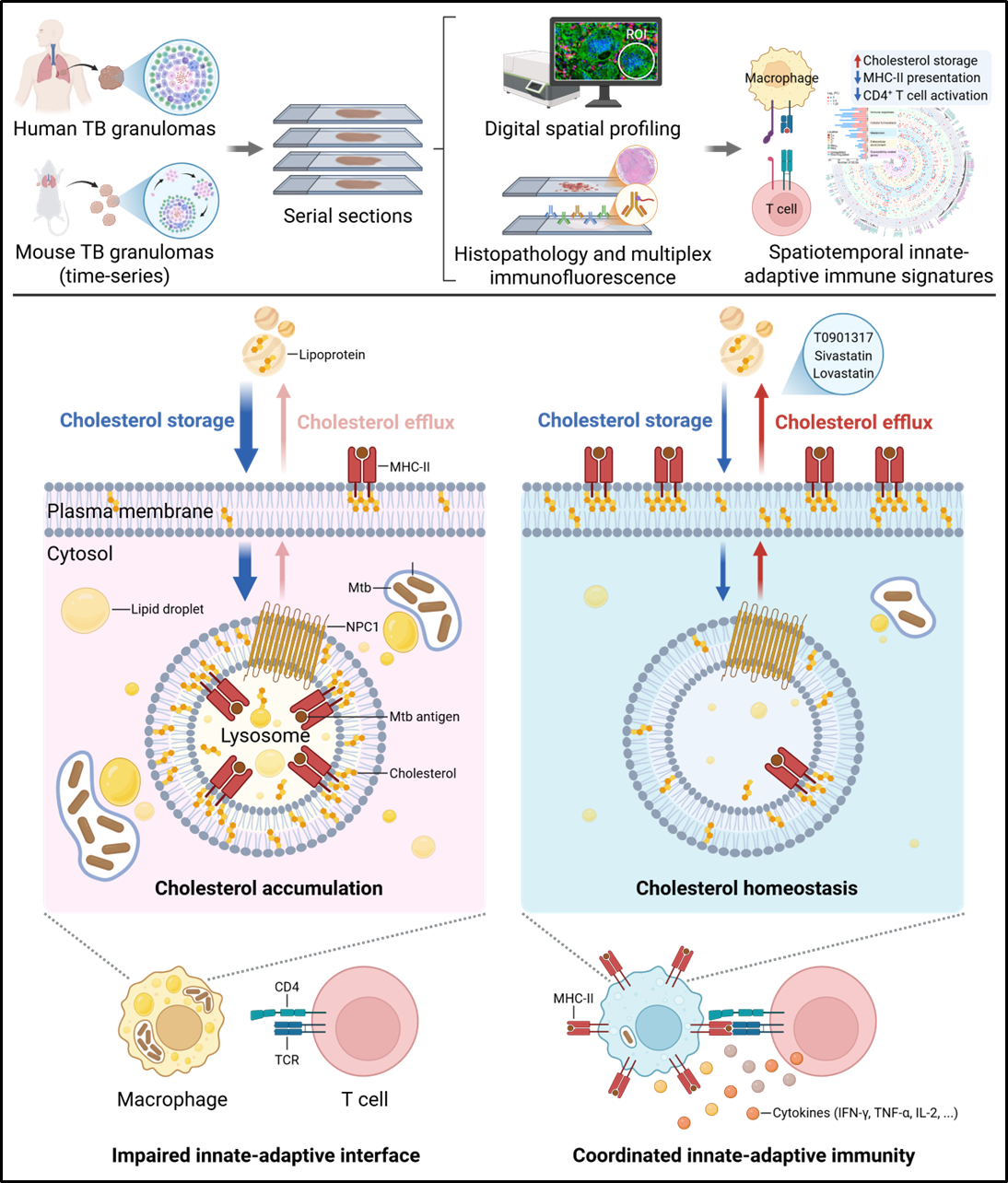
Figure 1. Schematic diagram of the mechanism by which Mtb induces lysosomal cholesterol accumulation to impair the macrophage-CD4+ T cell interaction (image by Prof. LIU Cui Hua’s group)
Full text links: Lipid accumulation in tuberculosis granulomas inhibits macrophage-CD4+ T cell interactions and infection control